I am not looking simply for a way to get rid of these culture tubes. I am looking to increase the number of amateur brewers who culture yeast. If I all wanted to do was sell the tubes, I would list them on eBay.
I would like part these tubes out in two dozen lots to brewers who are serious about learning how to isolate yeast and maintain a yeast bank. Maintaining a yeast bank is a hobby unto itself that requires an investment in time. Anyone who is solely looking to get into yeast culturing as a way to save money is going to quickly tire of the commitment that is required to maintain a healthy yeast bank.
Why am I limiting the lots to two dozen? Well, two dozen is really the smallest number of tubes that someone who is seriously interested in culturing should own. Yeast strains should be banked two tubes per strain or culture. One wants to have a back up in case a slant goes south, and one needs to have blank slants available for periodic subculturing because slanting is not a "set and forget it" way to maintain yeast. Two dozen culture tubes will allow a brewer to maintain up to about eight cultures, but six is more usual. It is difficult to prepare media for less than a dozen slants because one is only going to use about 3 to 4 milliliters of media per tubes. The tubes only hold 8ml, and one only fills the tubes 1/3rd to 40% full. I use a straight tip irrigation syringe to fill my tubes. This type of tip is known as a catheter tip.

I suggest heating the media up in a 2 to 4 cup glass measuring cup in a microwave. One only needs to heat the media until the agar melts. The pressure cooker will take care of rendering the media absolutely sterile.
Simple Solid Media
pale DME - 5% w/v
agar flakes or powder - 1.5% w/v
Solid MYGP
pale DME - 0.3% w/v
yeast extract - 0.3% w/v
soy-based peptone - 0.5% w/v
dextrose - 1.0% w/v
agar power - 1.5% w/v
One last thing, one should not transfer a liquid culture directly to slant. A liquid culture is plated for "singles" using a technique known as streaking. Well-isolated single colonies are transferred to slant.
Southern Tier's yeast strain in a 60mm x 15mm petri dish
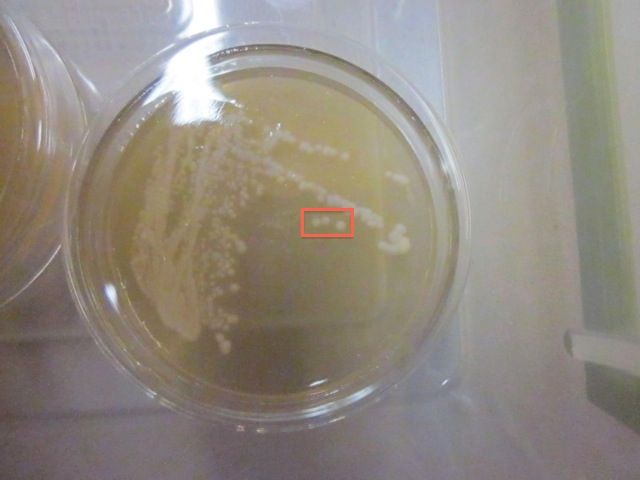
The well-isolated colonies (technically colony forming units, or CFUs) in the red box are each the offspring of a single yeast cell; hence, they are pure cultures.
Scottish and Newcastle's strain in 100mm x 10mm petri dish
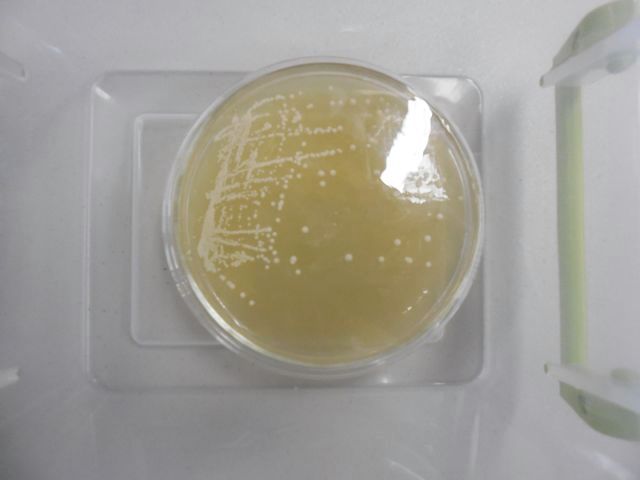
The colonies in the lower right hand quadrant are the candidates for transfer to slant.
There are several videos on YouTube that demonstrate how to streak plates and perform aseptic transfers over a flame source. I suggest starting out with a flint glass alcohol lamp.
I like this style the best (
http://prolabscientific.com/Flint-Glass-Alcohol-Lamps-p-18335.html)

Even a small Bunsen burner produces an intense flame that may be too much for someone who is just starting out. I would practice sterilizing the loop to get a feel for how it is performed before attempting a transfer. Like learning to ride a bicycle or roller skate, there is a learning curve with aseptic technique. However, just like learning how to ride a bicycle or roller skate, it is a set movements that one's hands remember even after long layoffs.
With the above said, while I am not looking to to make money on this venture, I am also not looking to lose money either. I need to look into shipping because I would like to find a way to bring the total cost down to around $35.00 including shipping.